Iso 13485 2003
The new standard ISO Medical device quality management system was published on March 1, 16 Compared with ISO , the requirements have changed significantly and organizations with ISO certification are now in a transition period.

Iso 13485 2003. It represents the requirements for a comprehensive quality management system for the design and manufacture of medical devicesThis standard supersedes earlier documents such as EN (1993. ISO Certification Sensortechnics Certifies Its Consultation, Design, Development and Production Processes According to ISO Sensortechnics is a leading company in the development and manufacture of high quality sensors, custom sensing solutions as well as fluidic control systems. Corresponding clause in ISO Cleanliness of product and contamination control In the new version of the standard, this clause is extended and the organizations are required to identify products that cannot be cleaned prior to sterilization or use.
BS EN ISO replaces BS EN ISO which has been withdrawn Who should buy it?. Equivalence ISO Superceding Superceded by LEGALLY BINDING DOCUMENT Step Out From the Old to the NewJawaharlal Nehru Invent a new India using knowledgeSatyanarayan Gangaram Pitroda Addeddate 1744 Identifier govinisiso Identifierark. The differences between ISO 900 and is that ISO 9001 is an international standard for a quality management system ISO 9001 standard is used to install the best possible format when ensuring consumer satisfaction with products and services On the other hand, ISO is the standard for a medical device quality management system.
This web page summarizes ISO 16 It highlights the main points It does not present detail To get the complete Plain English standard, please consider purchasing our Title 45 ISO 16 Translated into Plain English Title 45 is detailed, accurate, and complete. I would like to share eleven clauses that have significantly changed in ISO from ISO and how these changes relate to FDA 21 CFR Part 0 1 ISO CLAUSE 4 QUALITY MANAGEMENT SYSTEM & 41 GENERAL REQUIREMENTS The biggest change of these clauses against ISO is the 16 version requires application of a. Professional Plastics is a Master Distribution Partner to.
ISO ISO was released in March 16 All companies have 3 years in which to transition There are few industries where the importance of product conformity is as crucial as the design, manufacture, and testing of medical devices. This third edition of ISO cancels and replaces the second edition (ISO ) and ISO/TR , which have been technically revised It also incorporates the Technical Corrigendum ISO /Cor109 A summary of the changes incorporated into this edition compared with the previous edition is given in Annex A. OVERVIEW of ISO Medical In addition to Rochling's ISO certification, they are now also ISO Certified Röchling Engineering Plastics is the only manufacturer in the US to be ISO certified that supplies our broad product offering of engineering plastic stock shapes to the medical market;.
EN ISO ISO specifies requirements for a quality management system where an organization needs to demonstrate its ability to provide medical devices and related services that consistently meet customer requirements and regulatory requirements applicable to medical devices and related services The primary objective of ISO is to facilitate harmonized medical. Organizations dealing with the design, development, production, installation or servicing of medical equipment, devices and technology Why BSI?. Unfortunately, corrections on the ISO took a long time The new version of ISO was ready to be released in 16 although it relied on ISO So, when ISO was finally released with the new structure, was also already ready for release with the ISO structure.
ISO is a Management System Standard focused towards manufactures of Medical DevicesThe ISO Certificate was published by ISO in 03 The ISO standards outline a Comprehensive Management System for medical device manufacturers The ISO standard primarily facilitates medical device regulatory requirements that are. Currently, ISO and 16 will coexist which allows time for everyone to transition to the new standard ISO has an increased emphasis on regulatory requirements, risk management, validation/verification & design transfer, outsourced processes & supplier control, and feedback. ISO Certification Sensortechnics Certifies Its Consultation, Design, Development and Production Processes According to ISO Sensortechnics is a leading company in the development and manufacture of high quality sensors, custom sensing solutions as well as fluidic control systems.
ISO Certification is recognized as a worldwide quality certification specific to the Medical Device industry According the International Organization for Standardization, "ISO specifies requirements for a quality management system where an organization needs to demonstrate its ability to provide medical devices and related services that consistently meet customer and. Quality Management System for Medical Devices ISO 03 Certification ISO is an ISO standard, published in 03, that represents the requirements for a comprehensive management system for the design and manufacture of medical devices This standard supersedes earlier documents such as EN and EN (both 1997), the ISO. We are global, we’re independent and we’re a trusted service provider to 80,000 businesses.
ISO/TR ), Medical devices — Quality management systems — Guidance on the application of ISO 13 ISO , Medical devices — Application of risk management to medical devices 14 ISO , Guidelines for quaity and/or environmental management systems auditing 15. Upgrade from ISO It outlines the general requirements of ISO Since registration to ISO is a lengthy and detailed process, it is strongly suggested that firms seeking registration retain the services of a reputable consulting firm PERRY JOHNSON CONSULTING, INC. ISO Scope Statement (located at Canon Boulevard, Newport News, VA Contract manufacturer and testing of electronicbased medical device components ISO The standard specifies the requirements for an environmental management system that an organization can use to enhance its environmental performance.
ISO = ISO Medical Device Requirements ISO Medical Devices Quality Management System requirements for regulatory purposes is an ISO standard, originally published in 1996 This standard incorporated aspects of ISO Quality Management System, but is specific to the global medical device industry. Findings – Unlike ISO , ISO stresses the safety and efficacy of medical devices that are being produced For this reason risk management is an essential process that needs to be adopted into the ISO quality management system. ISO Medical devices Quality management systems Requirements for regulatory purposes is an International Organization for Standardization (ISO) standard published for the first time in 1996;.
ISO promotes a process approach when developing, implementing, and improving a QMS Requirements from Customers & Regulatory Authorities Valueadded activities Information Flow Slide 5 of 86 ISO – An Overview (KL, Malaysia, March 08) Gunter Frey & Hideki Asai.

Can Csa Iso 03 Rovers Medical Devices
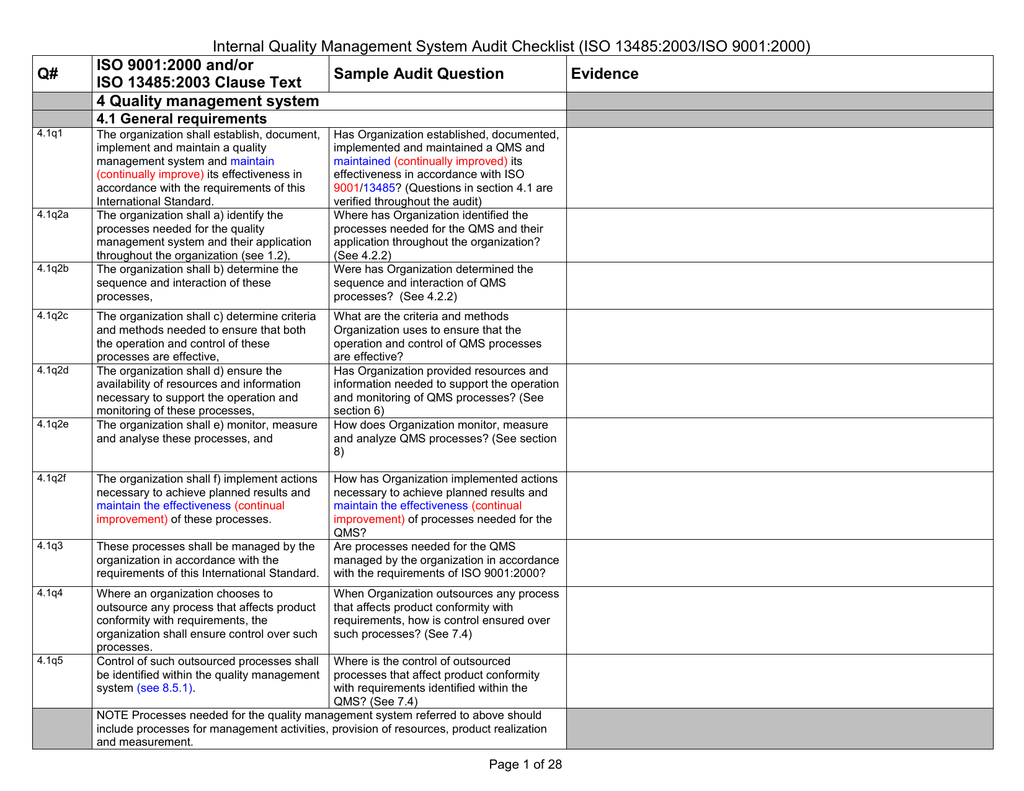
Internal Quality Management System Audit Checklist Iso 03 Iso 9001 00 Q

Iso 03 Ppunch Precision Punch And Tooling
Iso 13485 2003 のギャラリー

Certificate Iso 03 Globaler Rus Handel

Iso 16 Six Key Differences For Medical Device Companies

Fda 21 Cfr Part 0 Vs Iso 16 Vs Iso 03

Iso 03

Eclipse Aesthetics Receives Iso 03 Certification Biospace

Iso 03 Certification Aqlane Medical

Iso Iso 16 Awareness Training Tutorial Youtube

Certificate Iso 03 Canada Hettich

Leawell Iso 03 Medical Equipment Quality Management System Certificate Home Medical Equipment Product

Din En Iso Kirsch Pharmaceutical Refrigerators Blood Bank Refrigerators Blood Plasma Freezers Laboratory Refrigerators Laboratory Freezers

Ems And Odm News Info The Latest General Capabilities Information From Dbg

Iso 03 Medical Device Quality Management Royalty Free Cliparts Vectors And Stock Illustration Image 7268
Q Tbn And9gcsv9 Y3z3qq1xu0igpa0whkrbtvx Yloooj Zcne 2j4qsyfivi Usqp Cau

Iso 03

Litron Inc Receives Iso 03 Certification

Iso 03 Leader Life Camara Hiperbarica Medicina Hiperbarica

Softneta Is Certified By Iso Standard Iso 03 Softneta

Iso 03 Iso Quality Management

Iso Iqs International

Csoft Earns Iso 03 Certification For Medical Device Industry Csoft International
Q Tbn And9gcqggddjfniysqbq7tspizt5cxfzpifyswxndvtrs30 Usqp Cau

Premiere Resource Awarded Iso 03 Certification Medical Product Outsourcing

Iso 03 Leader Life Camara Hiperbarica Medicina Hiperbarica

Iso

Iso 16 New Revision Of Qms For Medical Devices Iso Consulting Services

Sustainability Iso 03 Mind Map

Airinspace Sup Sup Compliant With Iso 03 Standard Airinspace

Iso 03 Xenamed Industries

Welcome To Ibs Certifications Inc
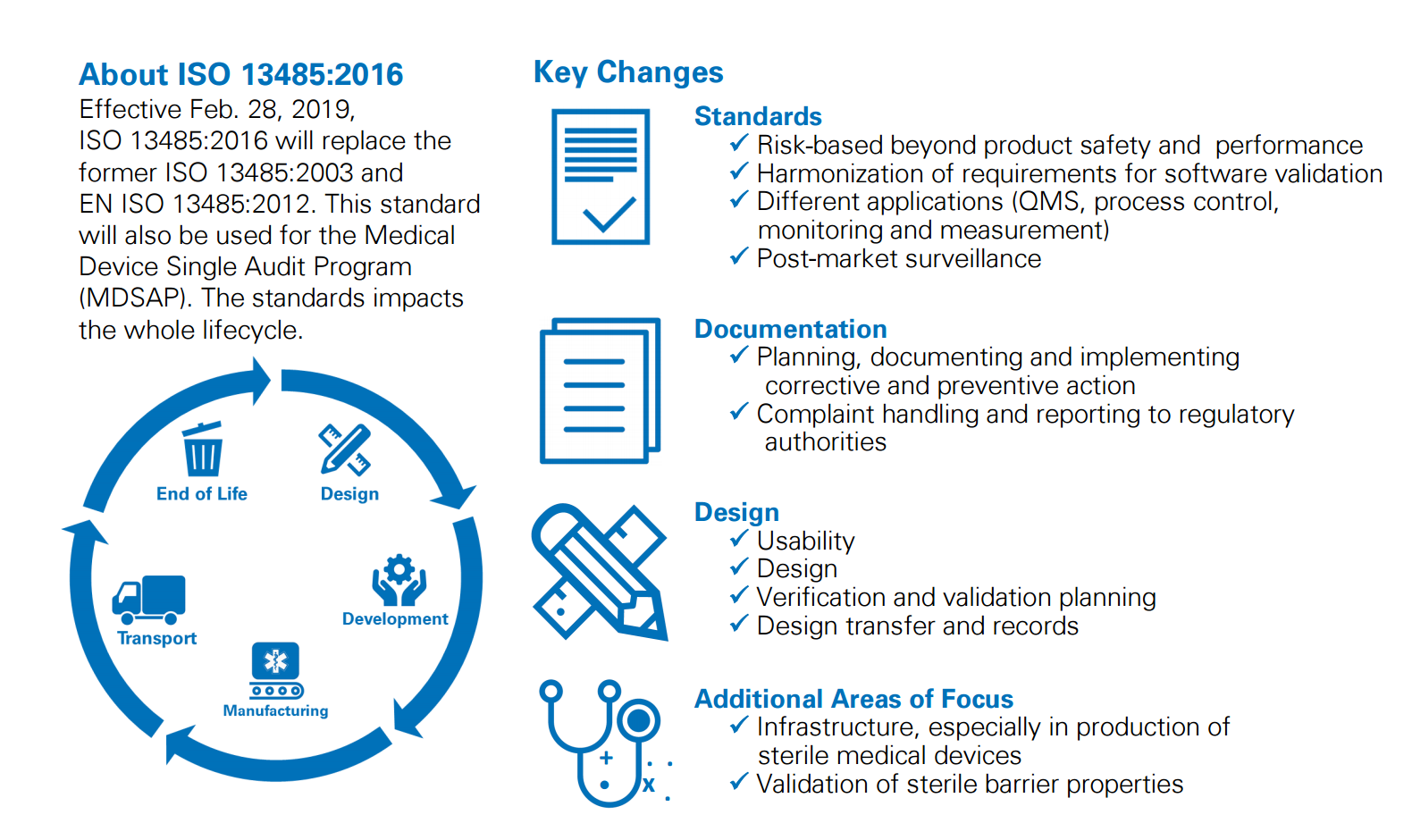
Publication Of Iso 16 Quality System Standard

Iso 03 Certificate 18 Philips Innovation Services

Iso Medical Devices Quality Management Systems Certification Bqai

Sn En Iso Medical Devices Quality Management Systems Snv

En Iso 03 Ac 07 Medical Devices Quality Management Systems Requirements For Regulatory Purposes Iso 03

Fristenkalender Zertifizierung Tuv Nord

Nuraleve Nordocs R D Facility Certified To The Iso 03 And Iso 9001 08

Ortho Baltic Conforms To Iso 03 Baltic Implants

Hs Design Received Bsi Recertification For Operating Qms That Complies With Iso 9001 08 Iso 03 Hs Design

Din En Iso 16 Update Was Ist Neu

En Iso 03 Ac 09 Compware Medical Gmbh

Zertifikat Iso 03 Ac 09 Heraeus Kulzer

Microvitae Technologies

Iso 03 Medical Devices Qms Certification In Chennai noor Global

Lemo Usa Achieves Iso 03 Certification

Cathetertechnology Certificate Iso 03 En

Iso Mdsap

Amazon In Buy Complete Iso 03 Quality Management System Book Online At Low Prices In India Complete Iso 03 Quality Management System Reviews Ratings

Delsys We Are Now An Iso 03 Facility Certifying That We Will Consistently Provide Products That Meet Customer And Regulatory Requirements T Co Z0ufq7eznb

We Are Iso 9001 08 Iso 03 Ns En Iso 12 Certified Asiagen Dengue Ns 1 Antigen Rapid Test Kit

Orthobaltic News

Kerecis Limited Receives Iso 03 Quality Management System Certification Kerecis

Transition Of Iso 03 To 16 Rx 360

Spellman S China Facility Receives New Iso Certifications

Iso 03 Certificate 18 Philips Innovation Services

Iso 03 Translated Into Plain English

En Iso 03 Medical Devices

Yamaha Motor Jw Wheelchairs Division Acquires Iso 03 Yamaha Motor Co Ltd

Iso 03

Iso Consultant Iso Certification Qms Iso Iso Medical

Iso 03 Quality Management System For Medical Device Certification Consulting Buy Iso Consulting Iso Consulting Medical Certification Product On Alibaba Com

Hui Awarded Medical Device Iso 03 Certification

Softneta Is Certified By Iso Standard Iso 03 Softneta

Iso 03 Certification Services In Andheri West Mumbai Beuna Vista Consultancy Certification Services Id

Interpretacao Iso 9001 08 Iso 03 E Rdc 16 13 Free Download Borrow And Streaming Internet Archive

Iso 16 Certification Qc Certification

Ofogh Sanat Medisa Company Achieved The Iso 03 Iso 9001 08 Quality Management System

Registrar Of Iso 03 Certificate In Bălcești Romania Iso 03 In Săliștea De Sus Romania Iso 03 Certification In Fierbinți Targ Romania Iso 03 Certification Registrar In Cavnic Romania Certification Service Of Iso

Iso 03 Abnova Has Just Renewed Its Iso 03 Certification News About Us Abnova

Iso 16 Certificate Iso 03 सर ट फ क शन सर व स आईएसओ 03 सर ट फ क शन सर व स आईएसओ 03 प रम णन स व ए In Nabha Patiala Revolutionary Consultants Id

Jcs Acquistive Infotech

Life Or Death In The Medical Industry Iso Matters Amax Blog And Insights

Iso 03 And Fda Qsr 21 Cfr 0 Internal Audit And Gap Analysis Checklist By Isoxpress

Iso 03 Quality Management Certification Timestrip

Iso 16 Revisions Webinar

Iso 03 Certification Services In New Delhi Delhi
Iso 16 New Terms To Take Note Of Qmswrapper

Onkocet

En Iso 12 Archives Mobile Shower Commode Chairs Raz Design Inc

A Comprehensive Guide To Iso Nqa

Iso 03 Certification Service In Kalighat Kolkata Q Matrix Consultancy Services

Iso Audit Guidelines

Ppt Transitioning From Iso 9001 08 To Iso 03 Going After Medical Device Business Powerpoint Presentation Id 6702

Iso 03 Certification Service In Sewak Park New Delhi Axxis Certification Id

Iso 03 First Sensor

Iso 03 Banners Standard Flags

Get Certification Iso Technomatical

What Are The Major Differences Between Iso 03 And The Latest Version Iso 16 Regarding Risk Managment And Company Leadership

Iso Medical Device Risk Management

Clontech Laboratories Inc Earns Iso 03 Certification

Quality And Certifications

Iso 16 Vs Iso 03

Pdf Iso 03 Implementation Reference Model From The Malaysian Smes Medical Device Industry
1
Iso Euro Certification

En Iso 03 Ac 07 Medical Devices Quality Management Systems Requirements For Regulatory Purposes Iso 03

Services Iso 03 Cmdms Certification In Offered By Pic Certification Private Limited Id

Bs En Iso Medical Devices Quality Management Systems Requirements For Regulatory Purposes

Cashido Hat Die Iso Zertifizierung Erhalten Cashido News Und Events Cashido Corporation

Iso 03 White Paper

Iso 03 Certification Definition What Is Iso 03 Find Iso 03 Companies On Thomasnet Com Supplier Diversity Quality Discovery



