Cell Line Development
The development of cell lines following a stable transfection step for the purpose of gene expression or recombinant protein production is an important method used by researchers Generating cell lines has often been inefficient and affected by inaccurate limited dilution methods or the shear stress caused by conventional flow cytometer and.

Cell line development. Biotherapeutics Cell Line Development Market Report provides an overview of the market based on key parameters such as market size, sales, sales analysis and key drivers The market size of the. A detailed study on the Cell Line Development Services Market published by Future Market Insights offers an indepth understanding of the various elements that are expected to influence the growth of the Cell Line Development Services Market in the upcoming years Further, by leveraging the data enclosed in the report, investors, stakeholders, established, and upcoming. Celltheon is the leader in Cell Line Development, Expression Technologies and Cell Engineering technologies The CELLTHEON SMART™ Platform has been used to date in 100 biotherapeutics for oncology, neurobiology and RA programs.
Latest developments and applications of transient expression systems used for large scale manufacture. Senior Director, Cell Line Development United States California Oceanside Gilead Sciences, Inc is a researchbased biopharmaceutical company that discovers, develops and commercializes innovative medicines in areas of unmet medical need. Stages of a typical cell line development process Transfection Transfect host cells with recombinant plasmids encoding protein of interest Selection of Pool of Transfected Cells Select cells that are stable and producing protein of interest Clonal Screening and Selection Screen and select clones for high expression of protein of interest.
New for , this year’s Cell Line Development & Engineering US meeting is colocated as part of BioProcess International US West, providing you with a unique opportunity to connect and learn from 1000 global scientists working further down the bioprocessing spectrum across upstream, downstream, analytics and commercial manufacturing. The Cell Line and Develoment Strategies conference will explore the many variables to be considered during the cell line development process, including verification and sequence analysis of the gene or protein of interest, codon optimization, vector construction and clone/host selection – a timeconsuming and expensive process. We understand that cell line development involves multiple, complex processes requiring consistency and reproducible results That’s why Gibco products are designed to provide you with integrated stable cell line development solutions to simplify your development workflow and accelerate your processes to commercial manufacture Request information.
Cell Line Development At Abzena, we see Cell Line Development as more than the process of developing high yielding, robust RCBs Our unique approach incorporates an upstream optimisation step within the CLD process, enabling accelerated downstream process development when linked to our GMP manufacturing facility in San Diego. Cell Line Development Get a custom iLite® cell line tailored to your needs iLite ® technology is a powerful cellbased technique used to measure and quantify molecules in biological samples It has applications throughout the drug development process as well as for monitoring biological drugs. Senior Director, Cell Line Development United States California Oceanside Gilead Sciences, Inc is a researchbased biopharmaceutical company that discovers, develops and commercializes innovative medicines in areas of unmet medical need.
New for , this year’s Cell Line Development & Engineering US meeting is colocated as part of BioProcess International US West, providing you with a unique opportunity to connect and learn from 1000 global scientists working further down the bioprocessing spectrum across upstream, downstream, analytics and commercial manufacturing. Ethical concerns surrounding abortionderived fetal cell lines in vaccines continue, even as several COVID19 vaccines are revealed to have been developed and/or tested using one or more of these aborted human fetal cell lines A list of each vaccine’s development and testing has been published by the Charlotte Lozier Institute (CLI). Based on the SUREtechnology Platform™ and unique worldclass expertise, Selexis SURE Cell Line Development™ Services significantly reduce the time, effort, and costs that are associated with the development of highperformance mammalian cell lines for therapeutic protein production (eg, development of monoclonal antibodies, bispecific monoclonal antibodies, growth factors, and enzymes).
The cell line development market is expected to grow with a CAGR of 132% from 1927 The report presents trends predominating in the global cell line development market and the factors driving the market and also the factors that act as hindrances. The Europe cell line development serum market is expected to reach US$ million in 27 from US$ million in 19 The market is estimated to grow with a CAGR of 5 9% from to 27. Gibco cell line development kits and services simplify your product development workflow and help you develop high quality production cell lines that will serve as the foundation for your product development for years to come.
The global cell line development market is anticipated to reach US$ 11, Mn in 27 from US$ 3, Mn in 18 The cell line development market is expected to grow with a CAGR of 132%. Even the most innovative R&D organizations still face significant data challenges in cell line development (CLD) To remain competitive, it is crucial to support collaboration and implement datadriven processes for the rapid creation of new high yield, reproducible cell lines Join us to learn how your teams can implement a modern informatics platform for efficient data management in cell. The Chinese hamster ovary (CHO) cell line is widely used for the production of therapeutic antibodies In a typical cell line development workflow, CHO cells are transfected with an IgG expressing vector A single CHO cell is obtained by limiting dilution cloning A high titer clone is selected using ELISA.
The Cell Line and Develoment Strategies conference will explore the many variables to be considered during the cell line development process, including verification and sequence analysis of the gene or protein of interest, codon optimization, vector construction and clone/host selection – a timeconsuming and expensive process. Polpharma Biologics offer their cell line development packages on a fee for service basis, this can either be as a stand alone cell line development project or as part of an integrated Idea to IND package We can also work on improving your current cell lines productivity with our SLIM™ or SPOT™ technology. Even the most innovative R&D organizations still face significant data challenges in cell line development (CLD) To remain competitive, it is crucial to support collaboration and implement datadriven processes for the rapid creation of new high yield, reproducible cell lines Join us to learn how your teams can implement a modern informatics platform for efficient data management in cell.
The market is estimated to grow with a CAGR of 5 9% from to 27 Increasing adoption of regenerative medicines, rising prevalence of cancer across the world, and increasing investments in R&D;. Based on the SUREtechnology Platform™ and unique worldclass expertise, Selexis SURE Cell Line Development™ Services significantly reduce the time, effort, and costs that are associated with the development of highperformance mammalian cell lines for therapeutic protein production (eg, development of monoclonal antibodies, bispecific monoclonal antibodies, growth factors, and enzymes). The Chinese hamster ovary (CHO) cell line is widely used for the production of therapeutic antibodies In a typical cell line development workflow, CHO cells are transfected with an IgG expressing vector A single CHO cell is obtained by limiting dilution cloning A high titer clone is selected using ELISA.
Cell Line Development At Abzena, we see Cell Line Development as more than the process of developing high yielding, robust RCBs Our unique approach incorporates an upstream optimisation step within the CLD process, enabling accelerated downstream process development when linked to our GMP manufacturing facility in San Diego. Cell culture has been a staple of biological and biomedical research since the inception of the first cell line, L929, or “ L cell ”, derived from mouse subcutaneous connective tissue by WR Earle in 1948 Only four years later, GE Gey published the immortalized HeLa line, the wellknown and widely used human cervical cancer cell line obtained from Henrietta Lacks. The MarketWatch News Department was not involved in the creation of this content Jan 14, 21 (Market Insight Reports) The Biotherapeutics Cell Line Development market report provides a.
Creating cell lines with future manufacturability, cost of goods and speed in mind is of critical importance in today’s biopharmaceutical development By working with our cell culture teams in our Slough (UK), Visp (CH), and Tuas (SG) sites, you will benefit from a recognized and stable cell line using our GS Xceed® Gene Expression System. Principal Scientist, Cell Line Development United States California Oceanside Gilead Sciences, Inc is a researchbased biopharmaceutical company that discovers, develops and commercializes. Cell Line Development At Abzena, we see Cell Line Development as more than the process of developing high yielding, robust RCBs Our unique approach incorporates an upstream optimisation step within the CLD process, enabling accelerated downstream process development when linked to our GMP manufacturing facility in San Diego.
We have an exciting opportunity for a Scientist of Cell Line Development to work with a team of doers working passionately in applying cuttingedge advances in cell editing and genomics. Bionova Scientific™ uses established mammalian expression systems to rapidly develop cell lines to generate Research Cell Banks (RCB) We also generate cell lines to support late stage process development to generate Master Cell Banks (MCB) suitable for cGMP manufacturing use. Cell Line Development Scientist/Senior Scientist (UK) We are a gene therapy company changing lives with every genetic advancement Asklepios BioPharmaceutical, Inc (AskBio) is a leading, clinicalstage gene therapy company founded in 01 based on the work of adenoassociated virus (AAV) gene therapy visionary and pioneer, Jude Samulski, PhD.
Principal Scientist, Cell Line Development United States California Oceanside Gilead Sciences, Inc is a researchbased biopharmaceutical company that discovers, develops and commercializes innovative medicines in areas of unmet medical need. Cell Culture Development Avioq has the experience, specialized facilities and mature Quality System to take your engineered mammalian and bacterial cell lines from the initial development phase to a highthroughput cGMP manufacturing process. Cell line Development Premas has deep experience and established labs and workflow on bacterial, yeast and mammalian expression platforms Our cell lines for development and clinical stage programs are high yield, wellcharacterised and compliant with the international regulatory guidelines (ICH).
Efficient HighQuality Cell Lines An efficient highquality cell line is the critical first step in any biopharmaceutical manufacturing process Achieving such a cell line can be a demanding task It requires precise craftsmanship, requiring both expertise and handson experience in process development as well as reliable technology platforms. A single Cell Dispensing Unit for Cell Line Development – taking the benefits of Limiting Dilution and combining with high seeding efficiencies June 07, 17 • Written by The Dish Introduction Identifying cell lines that have good manufacturing potential is critical to successful CHO cell based biomanufacturing. Gibco cell line development kits and services simplify your product development workflow and help you develop high quality production cell lines that will serve as the foundation for your product development for years to come.
Cell Line Development & Engineering Europe is a conference that covers topics such as Automation and high throughput technologies for cell line screening and clone selection;. Cell Line Development We offer efficient, flexible and high performing mammalian cell line development to transform your research into drug substances Leveraging the internal expression vector engineering expertise, we have optimized and engineered the proprietary vectors with selection stringency to be used with our proprietary host cell line. Biotherapeutics Cell Line Development Market Report provides an overview of the market based on key parameters such as market size, sales, sales analysis and key drivers The market size of the.
The Global Cell Line Development Market is expected to grow from JPY 410, Million in to JPY 764, Million by the end of 25 5\ The Global Cell Line Development Market is. Celltheon is the leader in Cell Line Development, Expression Technologies and Cell Engineering technologies The CELLTHEON SMART™ Platform has been used to date in 100 biotherapeutics for oncology, neurobiology and RA programs. The Global Cell Line Development Market is expected to grow from JPY 410, Million in to JPY 764, Million by the end of 25 5 The Global Cell Line Development Market is expected.
In Cell Line Development (CLD), you are looking for the rare, highproducing clones For the greatest genetic diversity, you need to screen thousands of cells The Opto CLD workflow screens thousands of clones with ease in less than a week Complex Antibody Molecules. 84 Bioprocess Cell Line Development jobs available on Indeedcom Apply to Scientist, Development Associate, Vice President of Business Development and more!. Cell Line Development We offer efficient, flexible and high performing mammalian cell line development to transform your research into drug substances Leveraging the internal expression vector engineering expertise, we have optimized and engineered the proprietary vectors with selection stringency to be used with our proprietary host cell line.
The development of cell lines following a stable transfection step for the purpose of gene expression or recombinant protein production is an important method used by researchers Generating cell lines has often been inefficient and affected by inaccurate limited dilution methods or the shear stress caused by conventional flow cytometer and. Cell Line Development Cell line development plays a major role in developing robust, costeffective biologic processes Drug developers need rapid availability of highperforming mammalian cell lines for therapeutic protein production, and flexibility. The Global Cell Line Development Market is expected to grow from JPY 410, Million in to JPY 764, Million by the end of 25 5 The Global Cell Line Development Market is expected.
In Cell Line Development (CLD), you are looking for the rare, highproducing clones For the greatest genetic diversity, you need to screen thousands of cells The Opto CLD workflow screens thousands of clones with ease in less than a week. Cell Line Development and Engineering Asia Novel, optimized, and innovative – with China’s growing and increasingly competitive biologics market, industry players are seeking out innovative and practical methods to develop a successful cell line and perfect production processes The 9 th Cell Line Development & Engineering Asia is the region’s leading conference for bringing together the brightest minds, innovators and industry players to uncover novel techniques, optimize workflows. Efficient HighQuality Cell Lines An efficient highquality cell line is the critical first step in any biopharmaceutical manufacturing process Achieving such a cell line can be a demanding task It requires precise craftsmanship, requiring both expertise and handson experience in process development as well as reliable technology platforms.
By pharmaceutical and biotechnology companies are the key factors driving the growth of the Europe cell line development serum marketNew York, Jan 15, 21 (GLOBE NEWSWIRE) Reportlinkercom. Biotherapeutics Cell Line Development Market Report provides an overview of the market based on key parameters such as market size, sales, sales analysis and key drivers The market size of the. Ethical concerns surrounding abortionderived fetal cell lines in vaccines continue, even as several COVID19 vaccines are revealed to have been developed and/or tested using one or more of these aborted human fetal cell lines A list of each vaccine’s development and testing has been published by the Charlotte Lozier Institute (CLI).
The Europe cell line development serum market is expected to reach US$ million in 27 from US$ million in 19. The North America cell line development serum market is expected to reach US$ million in 27 from US$ million in 19. Our stable cell line development activities are performed as standalone services or are fully integrated into larger scope process development programs Selected cell lines are used for the generation of KBI internal research cell banks (RCBs) For full scope process development and cGMP manufacturing programs, the master cell bank (MCB.
Biotherapeutics Cell Line Development Market Report provides an overview of the market based on key parameters such as market size, sales, sales analysis and key drivers The market size of the.

Cell Line Development Single Cell Sorting Molecular Devices

Speed Yield And Quality Critical For Cell Line Development Says Atum

Rapid Development Of High Quality Robust Mammalian Cell Culture Manufacturing Processes Bioprocess Internationalbioprocess International
Cell Line Development のギャラリー

Towards Next Generation Cho Cell Line Development And Engineering By Systems Approaches Sciencedirect

Improved Speed To Clinic By Increasing Automation In Cell Line Development Biooutsource
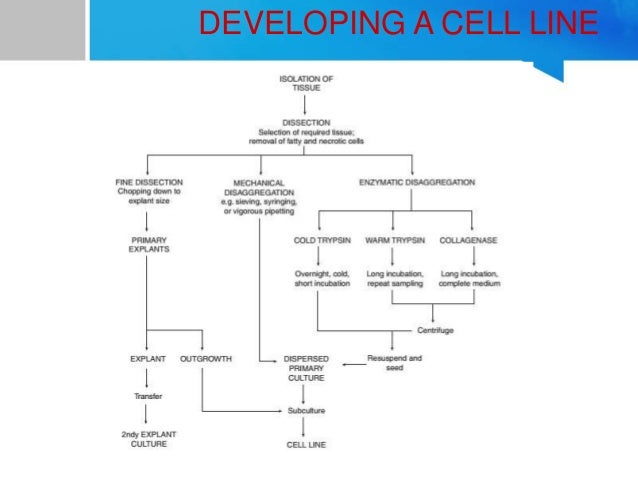
Cell Line Development Ol
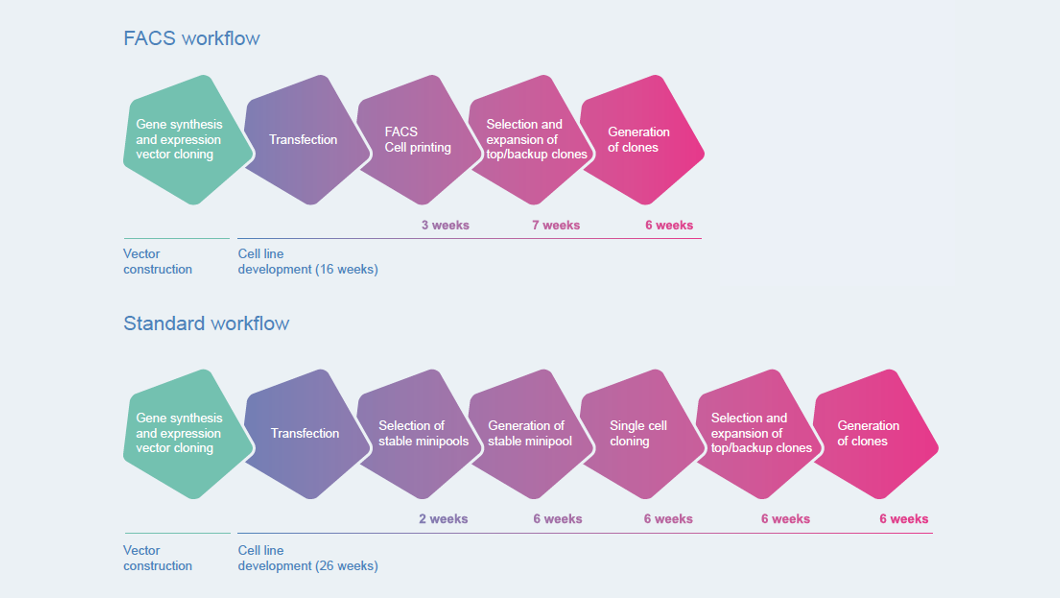
Cutting Time To Market For High Quality Therapeutics European Biotechnology

Cell Line Development

Cell Line Development Services Stable Cho Cell Line Development Cellca Biooutsource
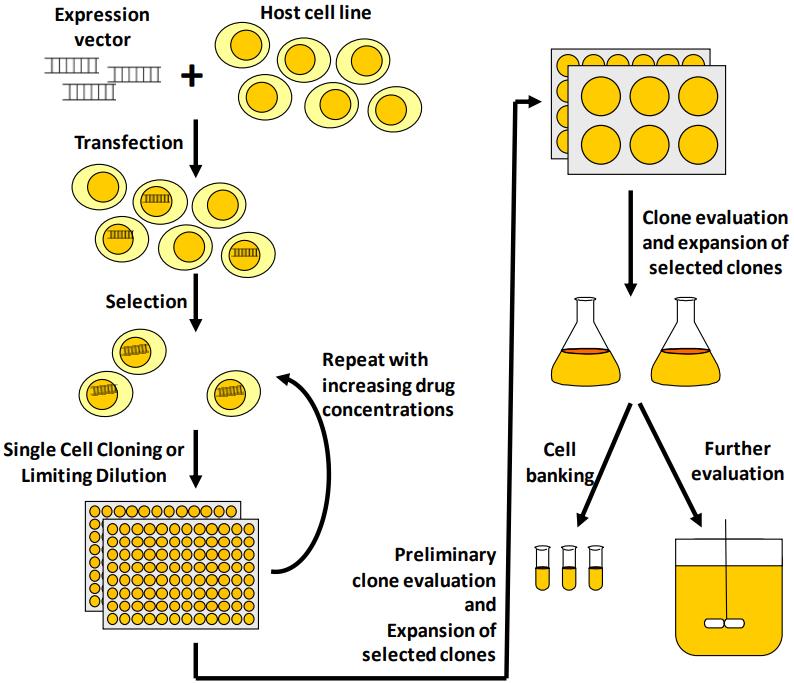
Stable Cell Line Construction Services Creative Biolabs

Best In Class Cell Line Development And Biologic Manufacturing Coming Together Selexis Sa
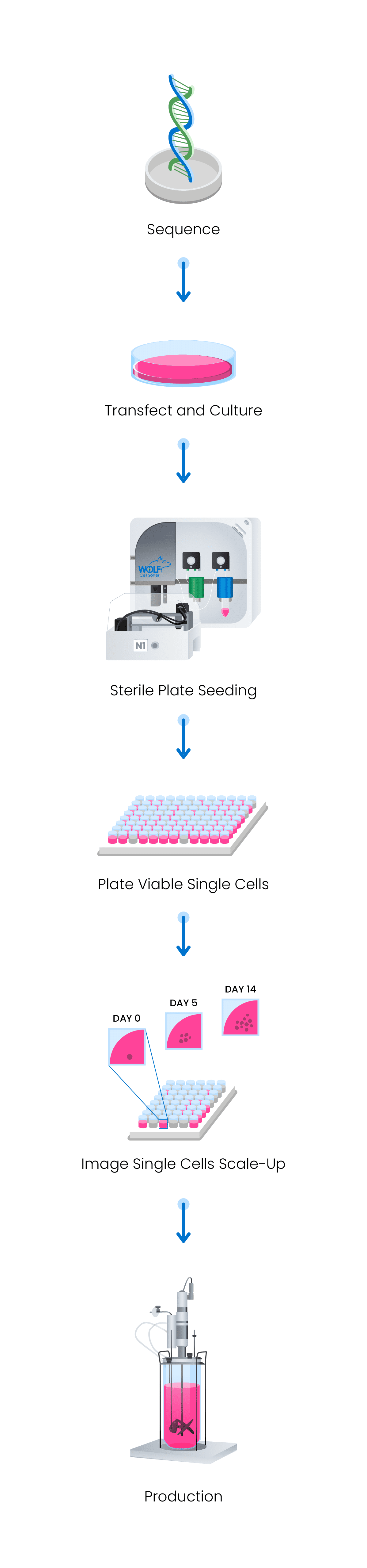
Cell Line Development Engineering Nanocellect

Stable Cell Line Cellpower For Assay Development
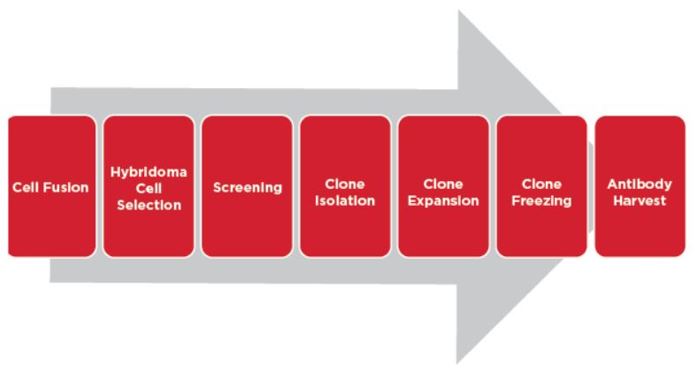
Automating The Cell Line Development Workflow Beckman Coulter

Rapid Cell Line Development With Integrated Protein Analysis Bioprocess Internationalbioprocess International

Cell Line Development Sciencedirect

Site Specific Integration Ushers In A New Era Of Precise Cho Cell Line Engineering Abstract Europe Pmc
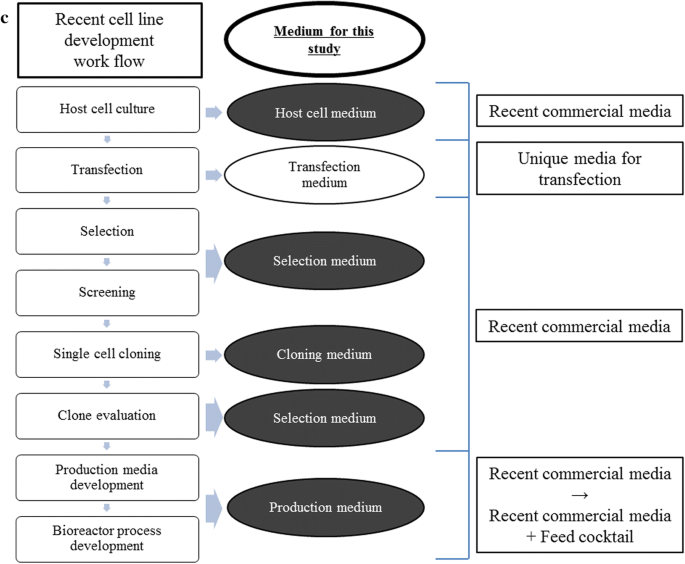
Rich Production Media As A Platform For Cho Cell Line Development Amb Express Full Text
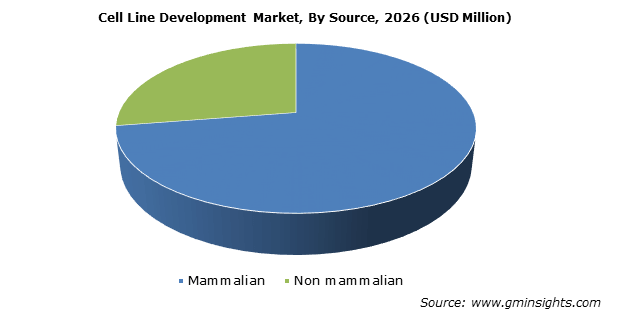
Cell Line Development Market Share Industry Outlook 26

Cell Line Development Ol

Cell Pool Generation And Cell Line Development For Pyc2 Engineering Download Scientific Diagram

Overview On The Cell Line Development Process Used In This Study Download Scientific Diagram

Cell Line Development Workflow Molecular Devices

Integrated Services Cell Line Development Gmp Fill Finish Kemwell
Automating Cell Line Development Labplan
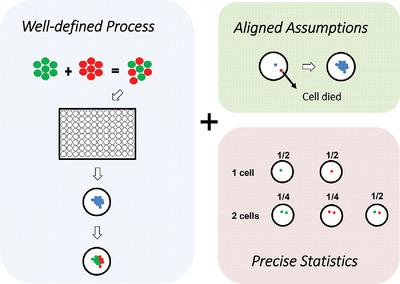
Methods For Estimating The Probability Of Clonality In Cell Line Development Biotechnology Journal X Mol

Cell Line Development Cld 2 0 Biooutsource
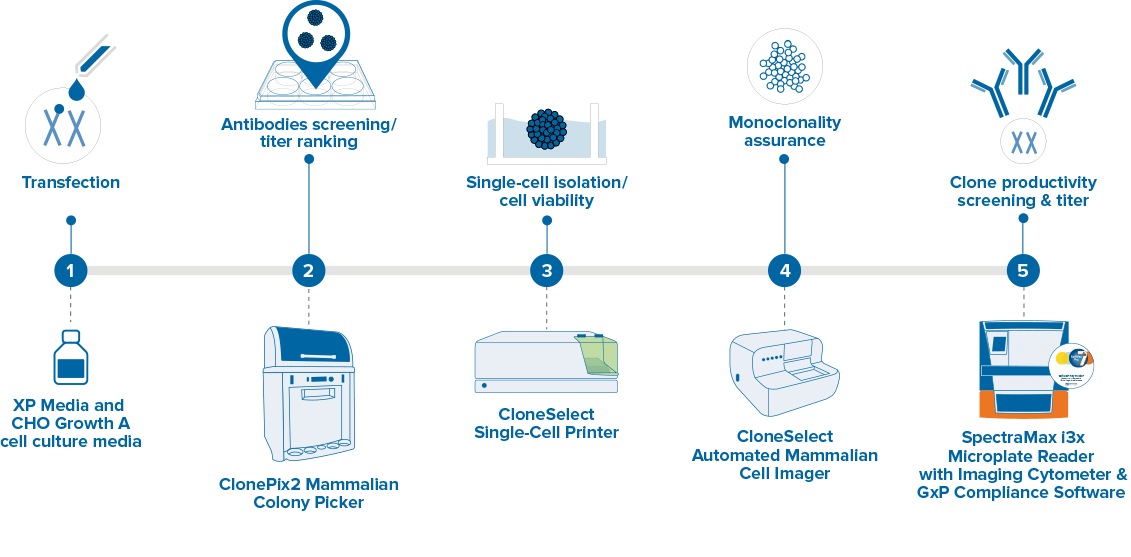
Cell Line Development Single Cell Sorting Molecular Devices
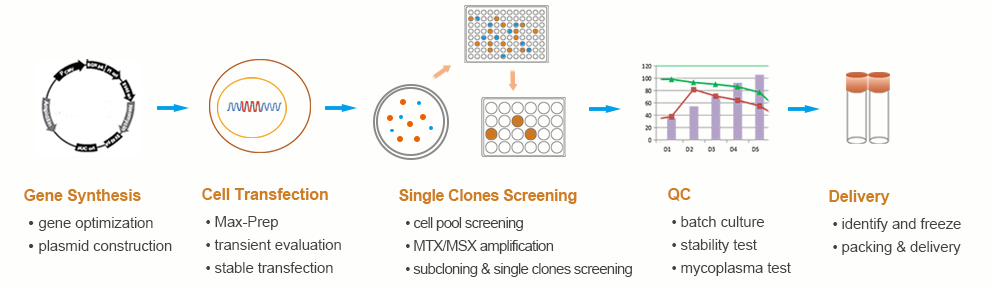
Stable Cell Line For Protein Production Biologicscorp
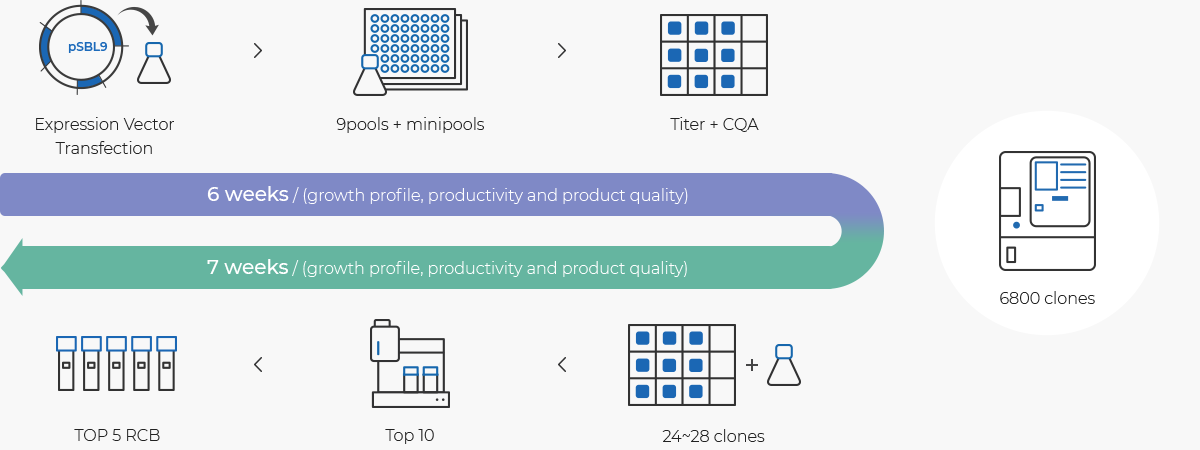
Cell Line Development Cdo Our Services Samsung Biologics
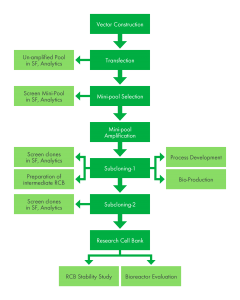
Stable Cell Line Development Kemp Proteins

North America Cell Line Development Market Size Share Report 24

Cell Line Development Accelerating Antibody Discovery By Monitoring Titer And Glycosylation With The Octet Platform White Paper Technology Networks
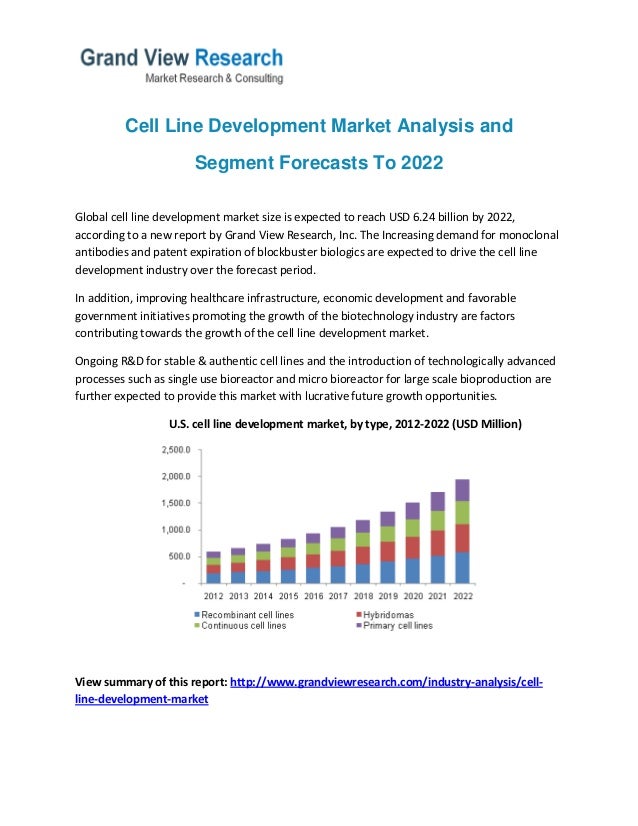
Cell Line Development Market Analysis And Growth
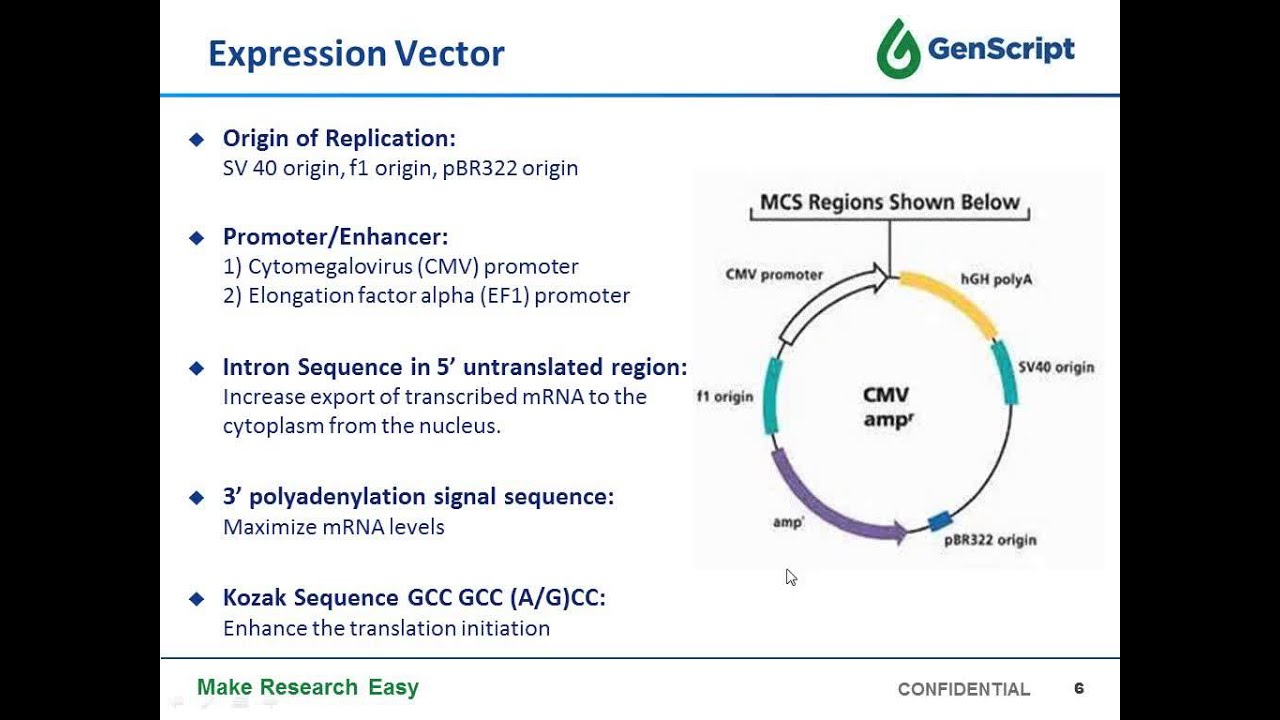
Stable Cell Line Development For Large Scale Antibody And Protein Production Youtube

Biodevelopment Services Process Development Celonic
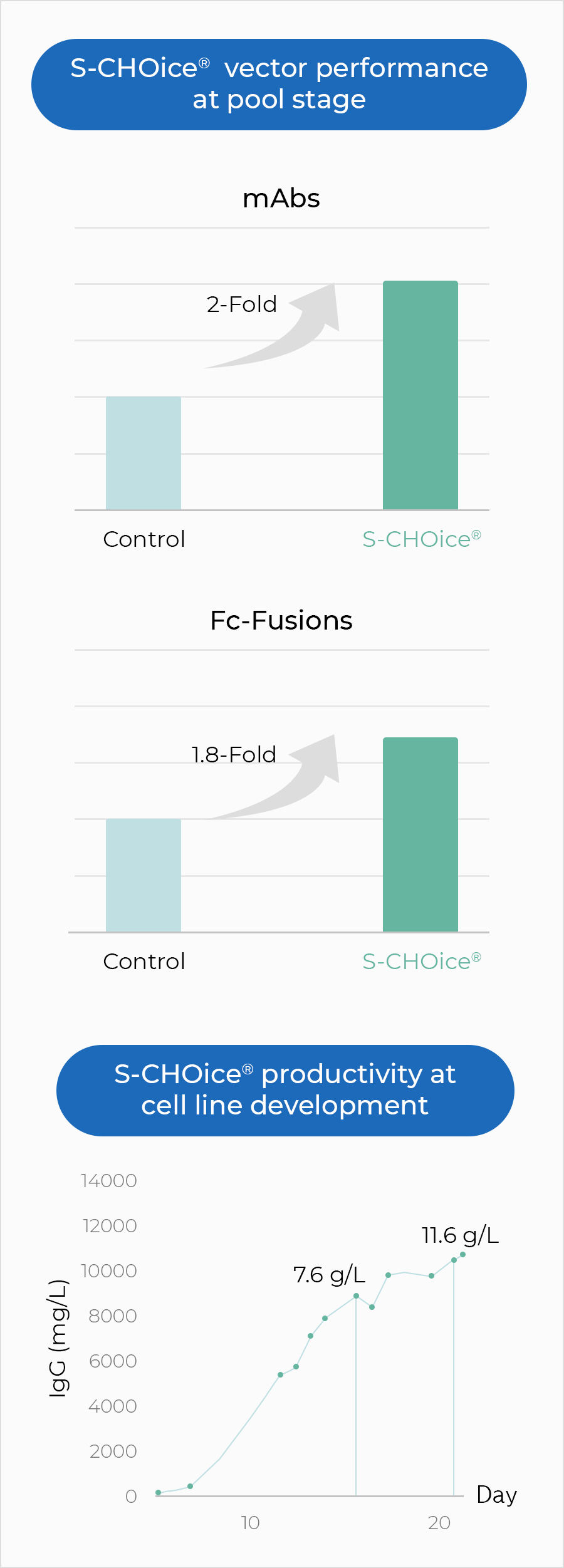
Cell Line Development Cdo Our Services Samsung Biologics
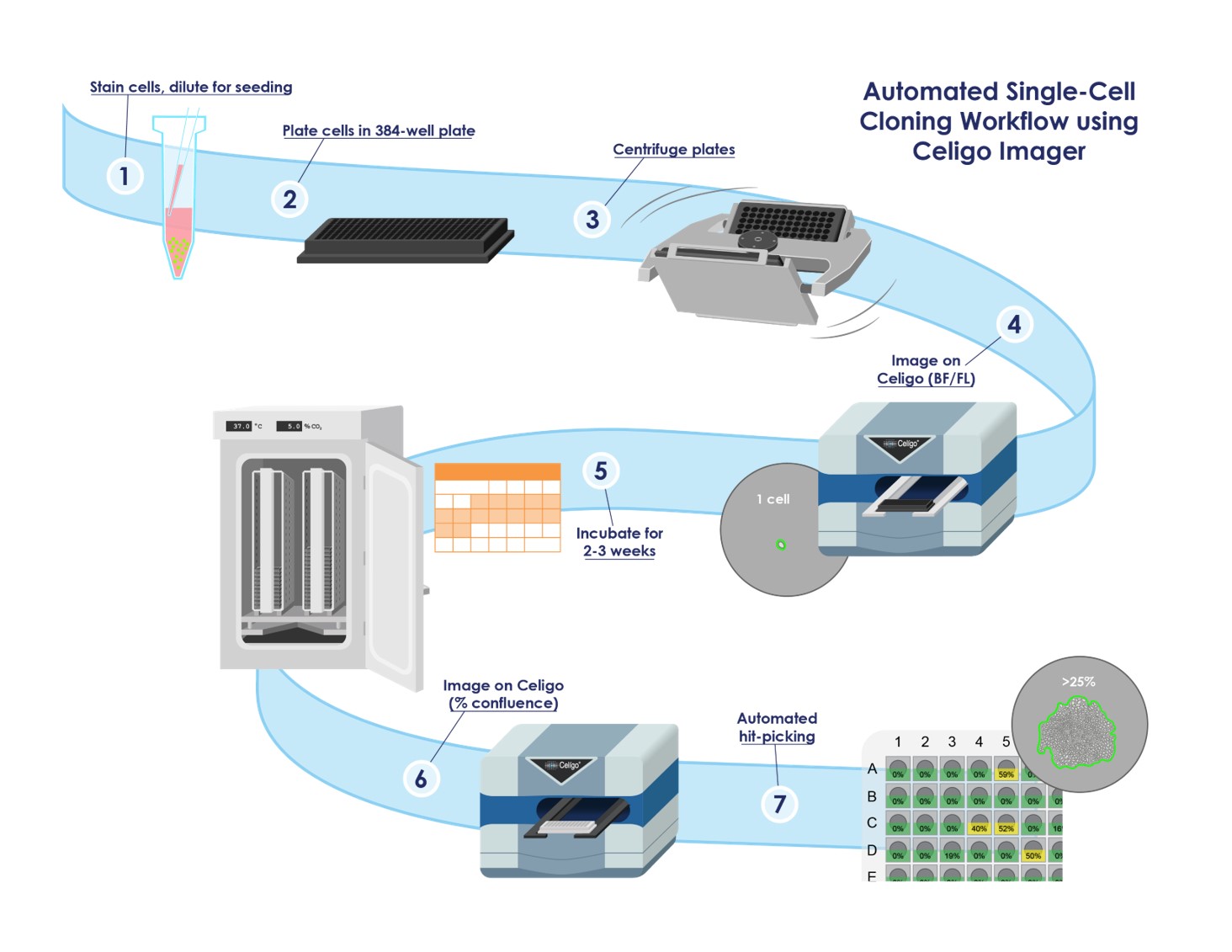
Modernize Your Cell Development Assays With High Throughput Image Cytometry

Cell Line Development
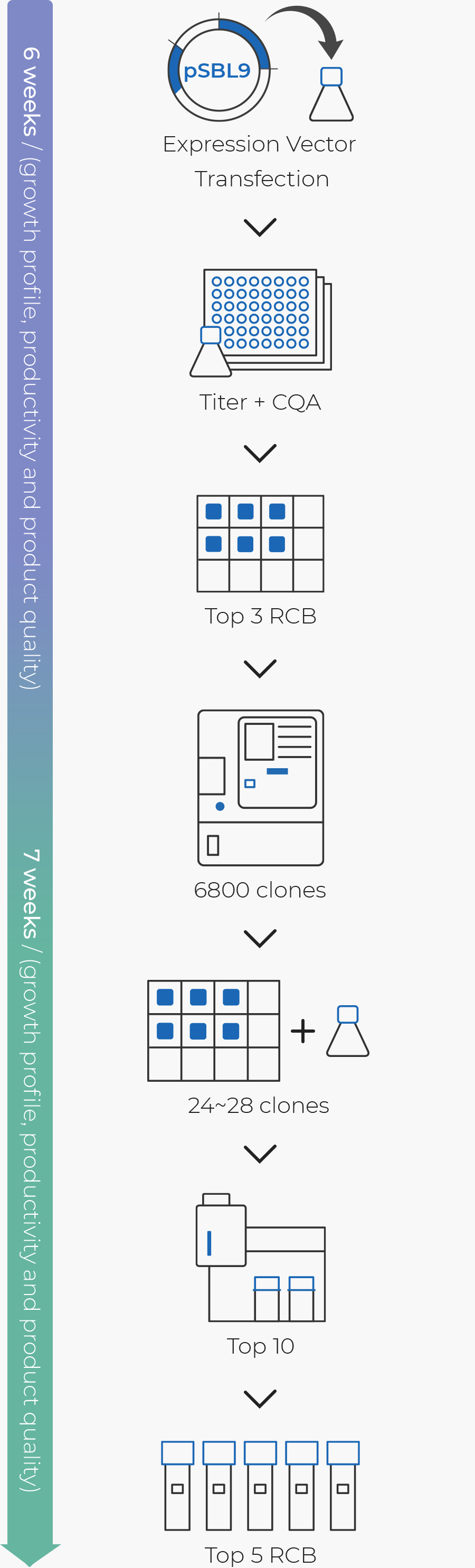
Cell Line Development Cdo Our Services Samsung Biologics

Cho Stable Cell Line Development

Get To Ind Faster Accelerated High Performance Cell Line Development Youtube

Cell Line Development Angel Bioscience Inc

Stable Cell Line Antibody Production Ab Scale Up Services Abnova
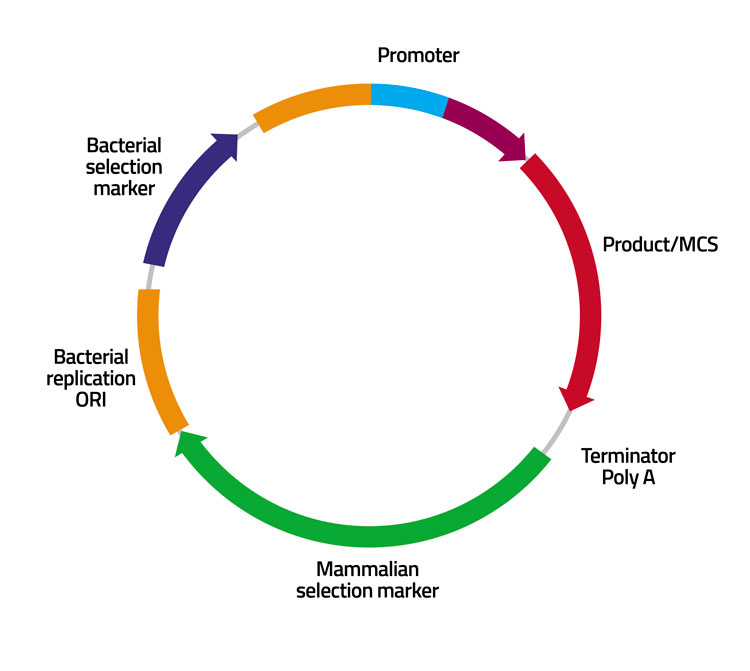
Cell Line Development Biologics Development From Early Phase To Ind

Solutions Bioprocess Development Turbocell Mammalian Expression Platform Rentschler Biopharma

Cell Line Development Trends Contract Pharma
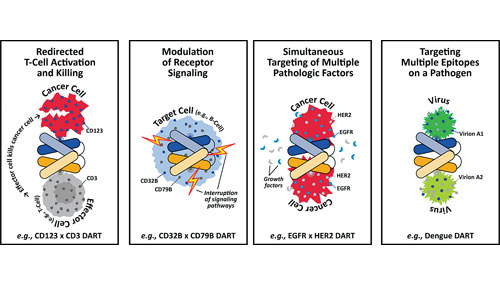
Cell Line Development Where Nature Meets Nurture

Stable Cell Line Cellpower For Assay Development

Cell Line Development Nexcelom Bioscience
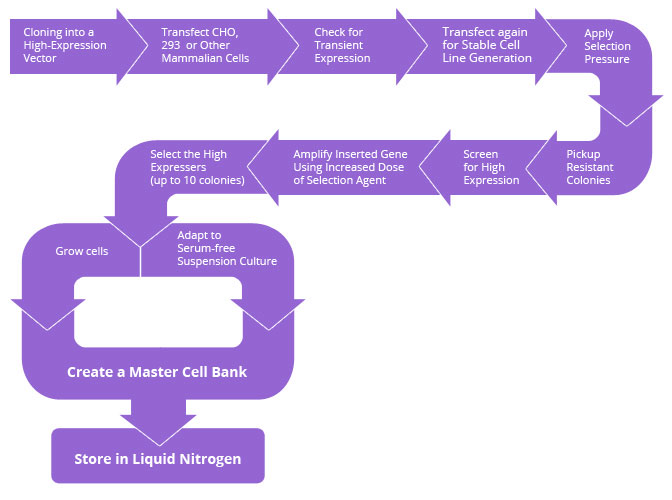
Stable Cell Line Development Cell Based Assays Derived Productions
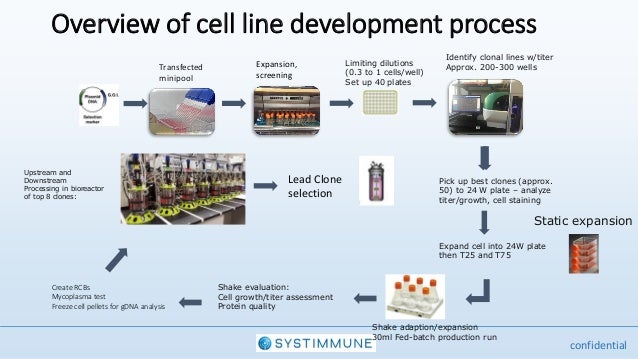
Presentation By Systimunne At The Cell Line Development Engineeri
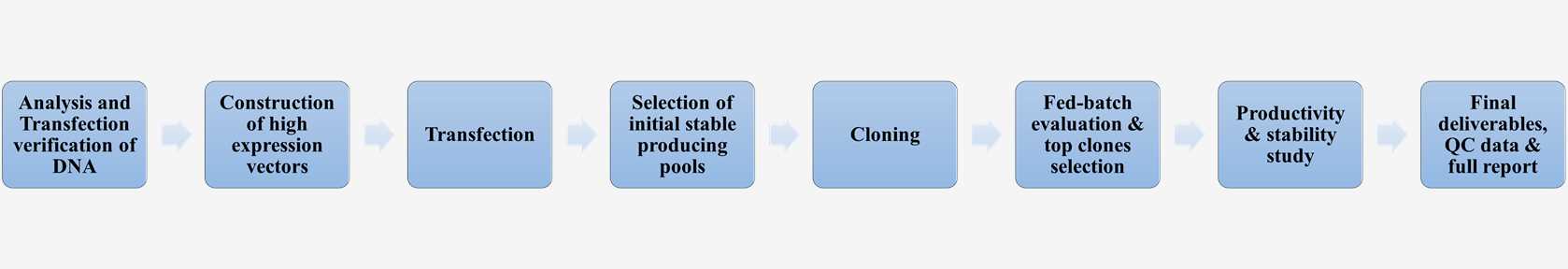
Cgmp Stable Cell Lines Creative Biolabs
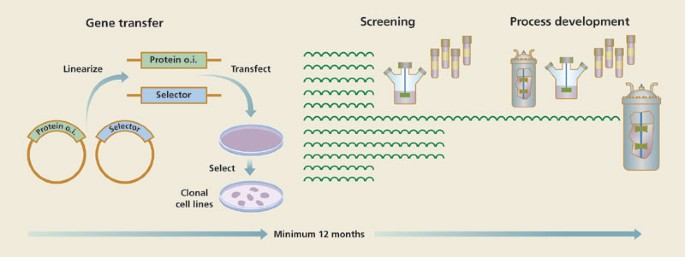
Production Of Recombinant Protein Therapeutics In Cultivated Mammalian Cells Nature Biotechnology
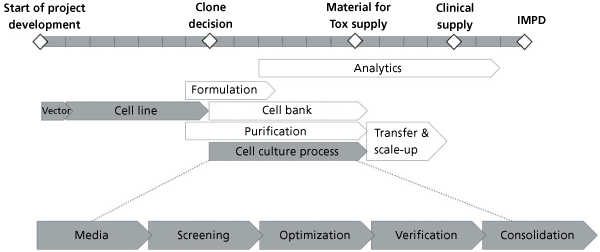
Platform Approach Speeds Process Development Bioprocess Development Forum

Overview Of Pbg Cell Line Development Download Scientific Diagram

Cell Line Development 101

Streamline The Workflow For Measuring Igg In Cell Line Development Molecular Devices
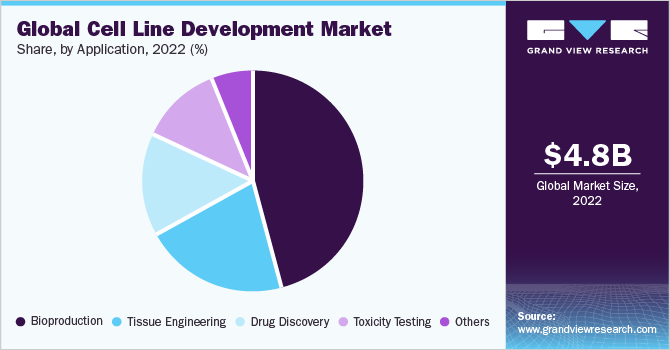
Cell Line Development Market Size Share Industry Report 12 22

Cell Culture Media And Feeds For Biopharmaceutical Production
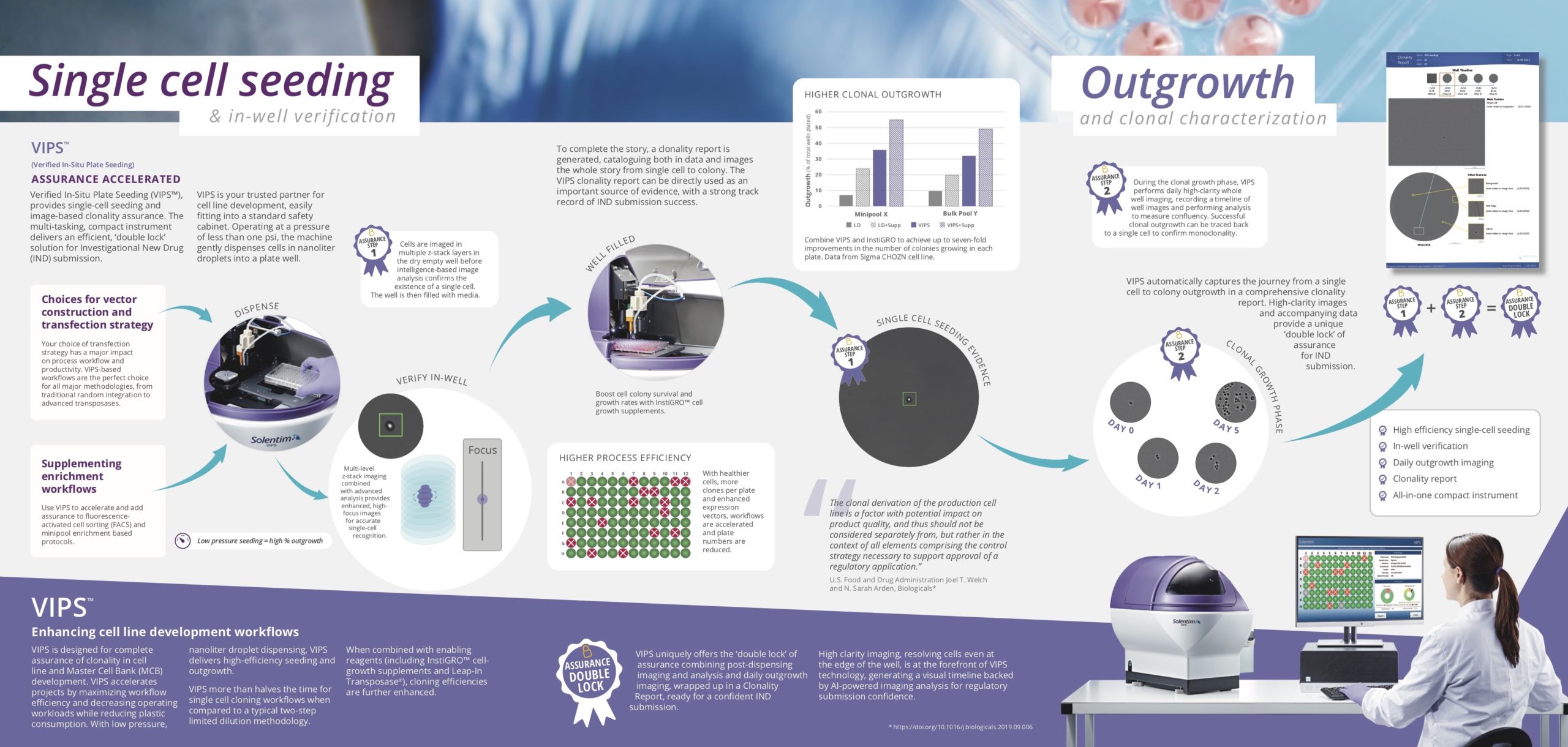
Double Lock Assurance Solentim
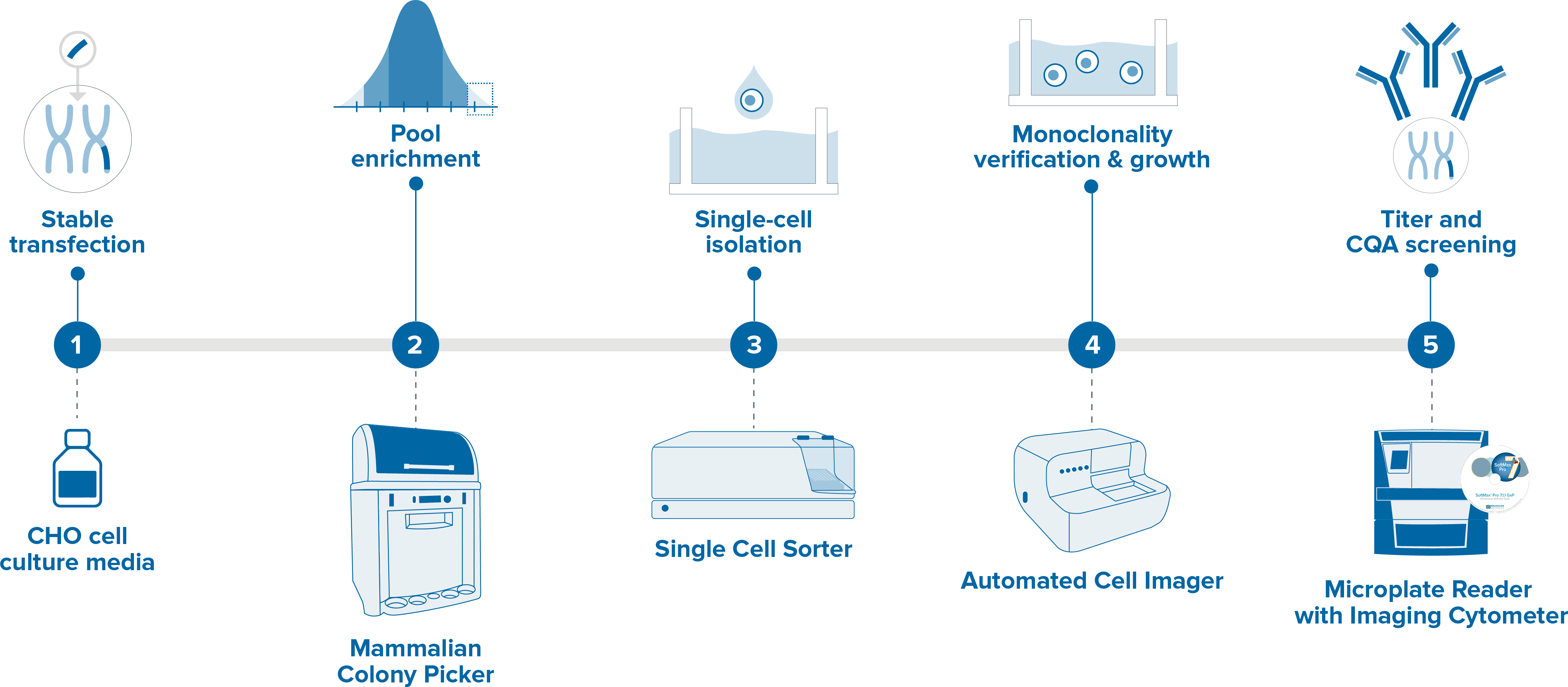
Stable Cell Line Development For Antibody Production Molecular Devices

Cell Line Development Sciencedirect

Cell Line Development Gtp Technology

Cell Line Development

Accelerating Timelines By Integrating Cell Line Development And Manufacturing Programs Bioprocess Internationalbioprocess International
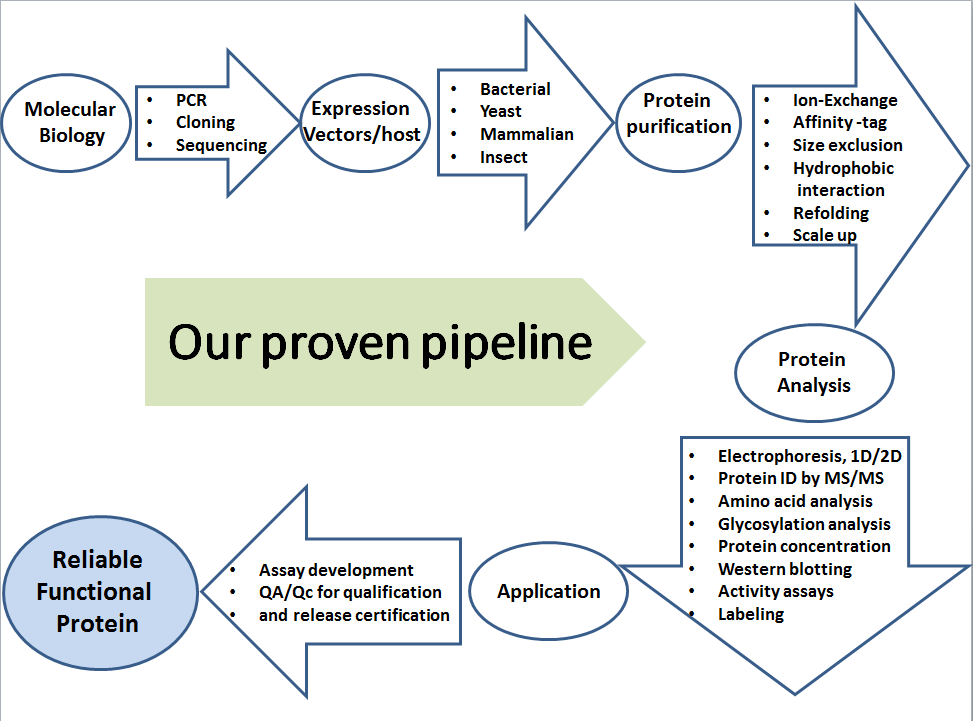
Cell Line Development Cell Development Facility Nebraska
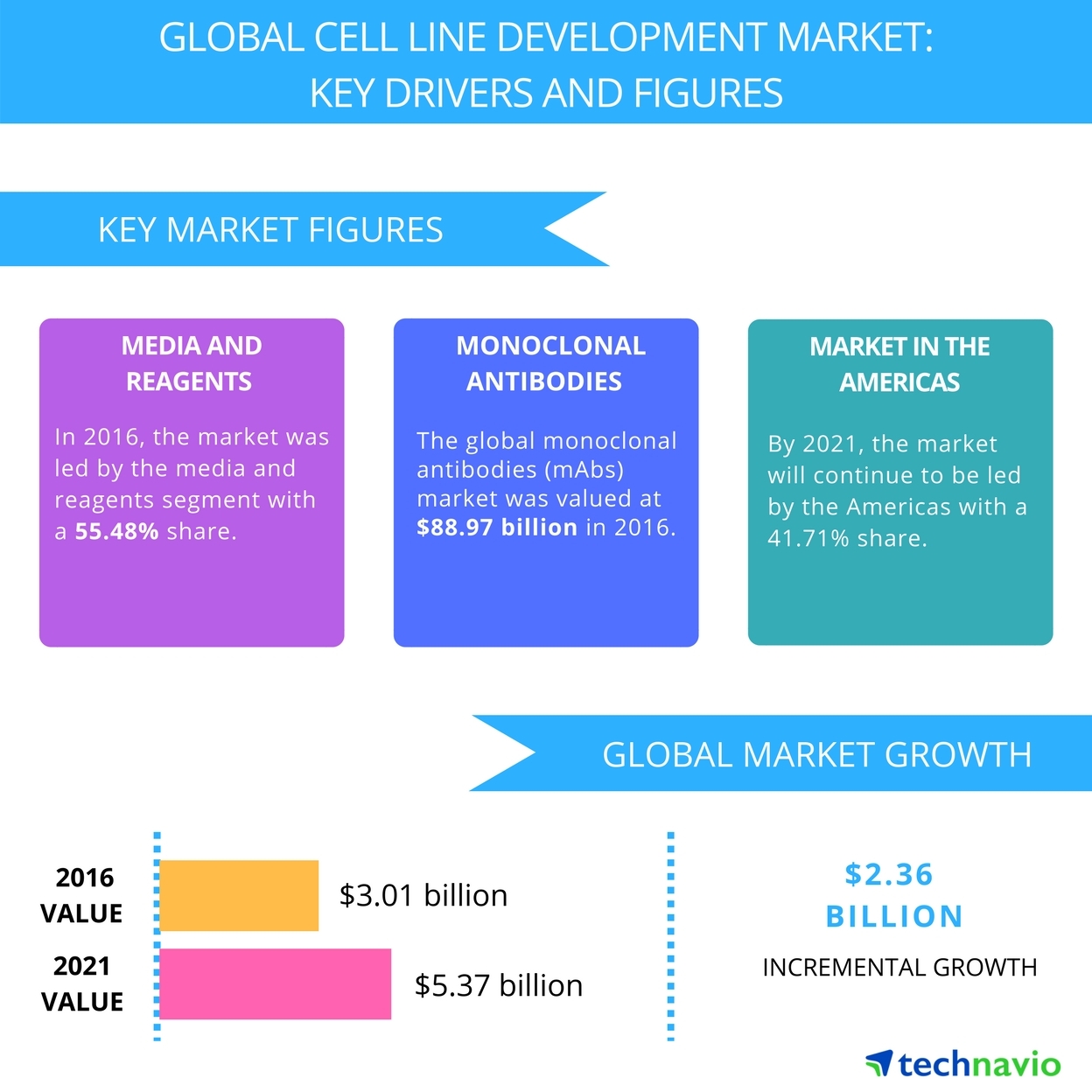
Global Cell Line Development Market Key Drivers And Forecast From Technavio Business Wire

Cell Line Development Berkeley Lights
Cell Line Development Services Cho Hek Excellgene

Fast Tracking Cell Line Development Biocompare The Buyer S Guide For Life Scientists
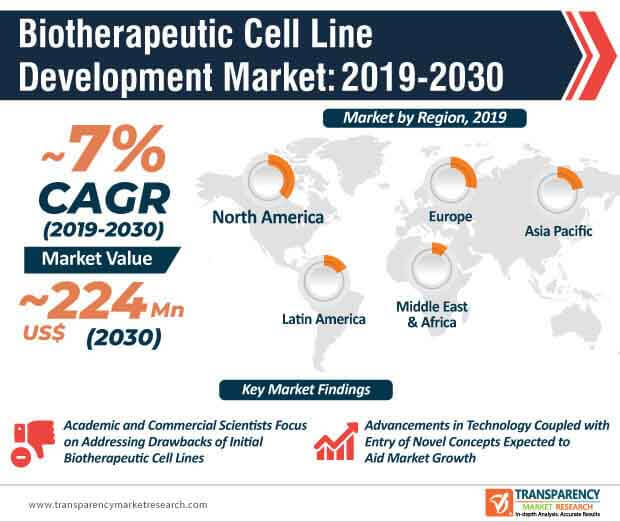
Biotherapeutics Cell Line Development Market By Process Geography And Forecast To 30 Tmr
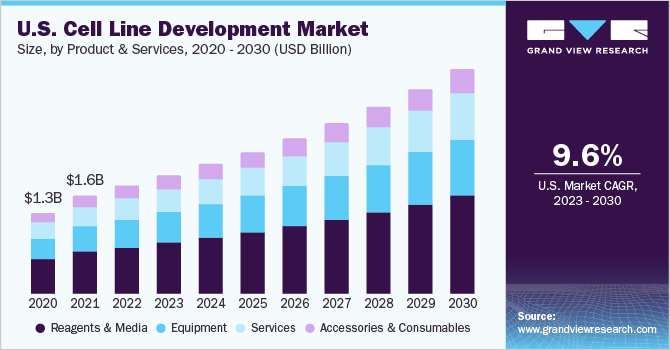
Cell Line Development Market Size Share Industry Report 12 22

Cell Line Development Namocell

Cell Line Development Services Stable Cho Cell Line Development Cellca Biooutsource

Overview On The Cell Line Development Process Used In This Study Download Scientific Diagram
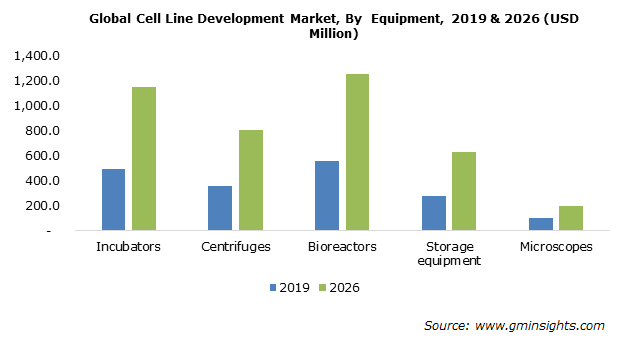
Cell Line Development Market Share Industry Outlook 26
Pro Active Cho Cell Line Development For Bioproduction Youtube
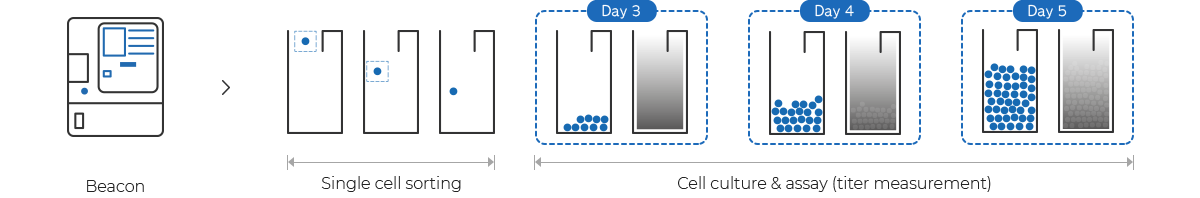
Cell Line Development Cdo Our Services Samsung Biologics
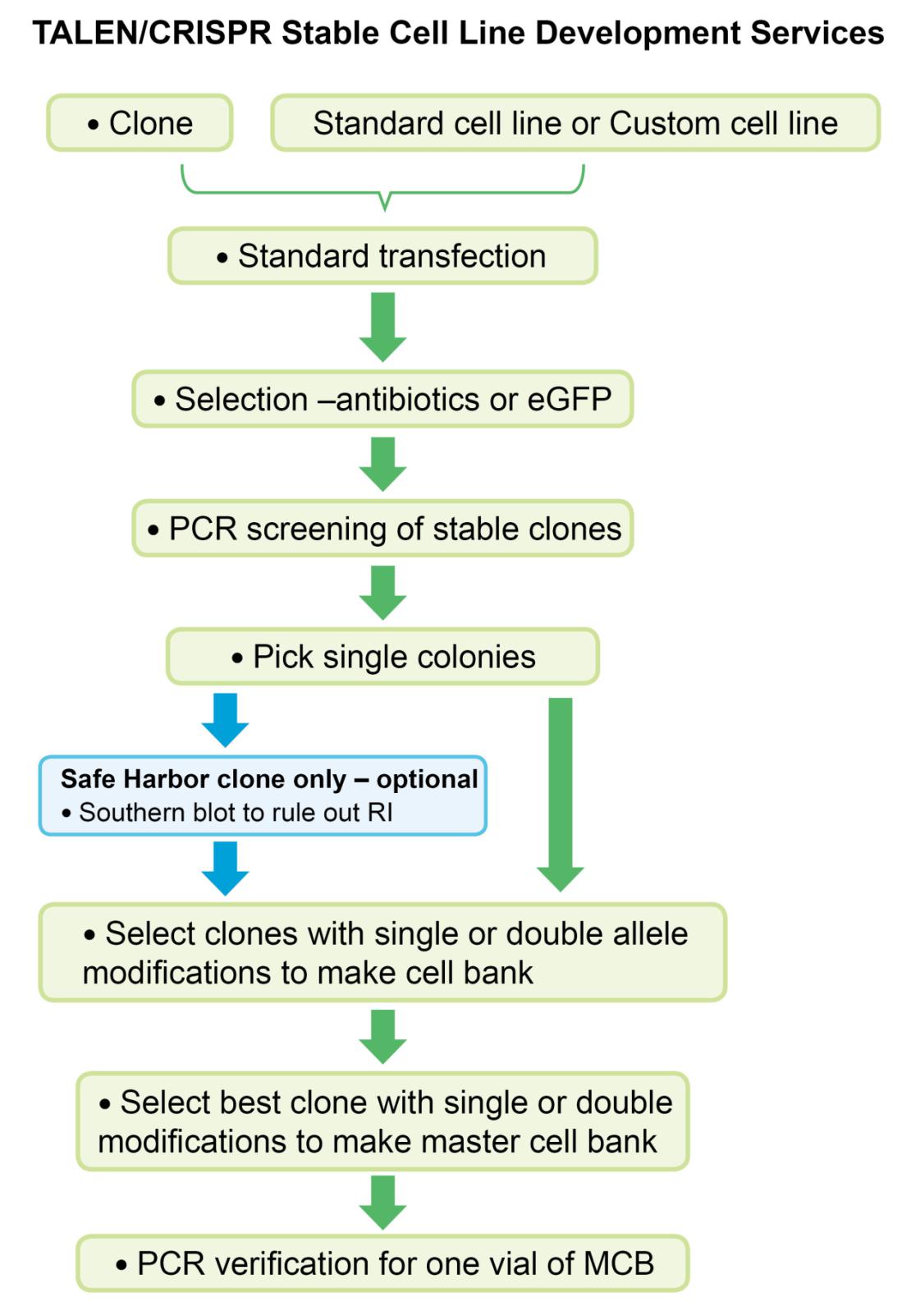
Ge Stable Cell Line Development Services Genecopoeia

Cell Line Process Development Jhl Biotech Inc

Apollo X Mammalian Expression Fujifilm Diosynth Biotechnologies

Cell Line Development

Mammalian Cell Line Development Gtp Technology

Cell Line Development Services Global Market

Cell Line Development Market Share Industry Outlook 26
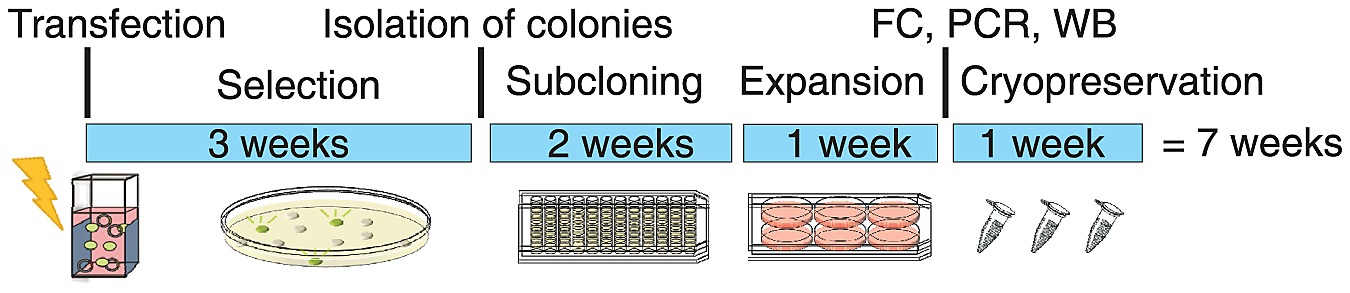
Timeline For Rfp Cell Line Development By Rmce
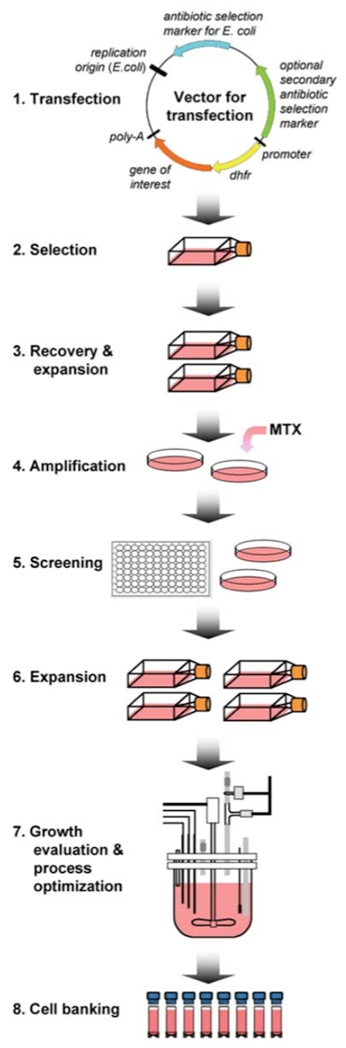
Recombinant Protein Expression In Mammalian Cells Hek293 Cho Profacgen
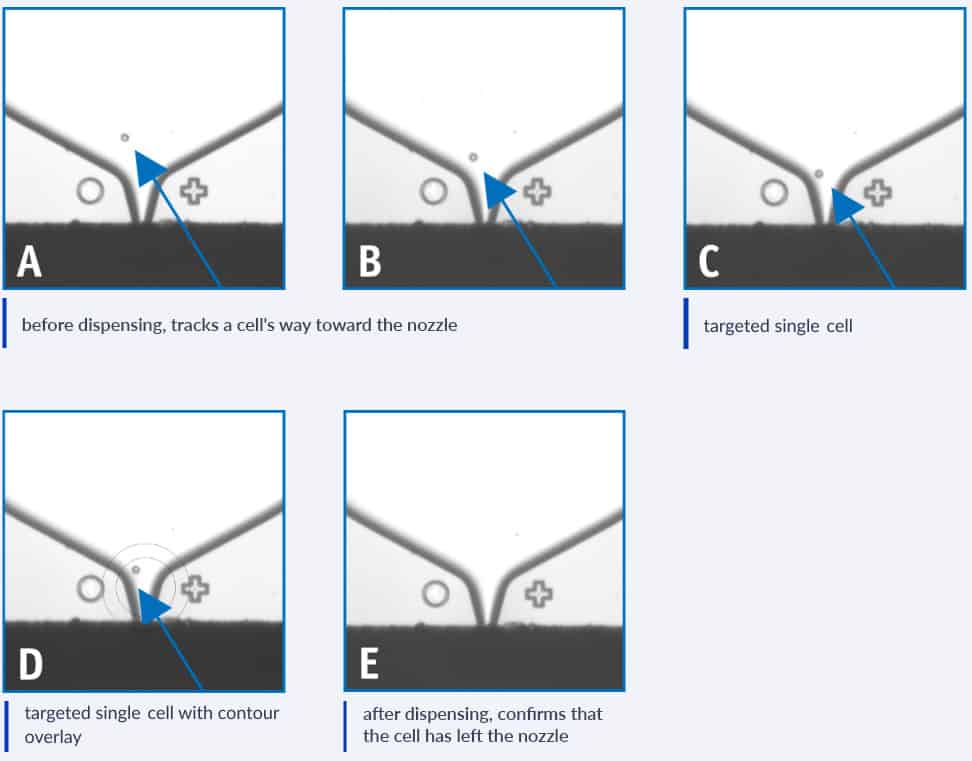
Cell Line Development Cytena

Automated Solutions Cell Line Development Beckman Coulter

Mammalian Cell Lines Monoclonal Antibody Development
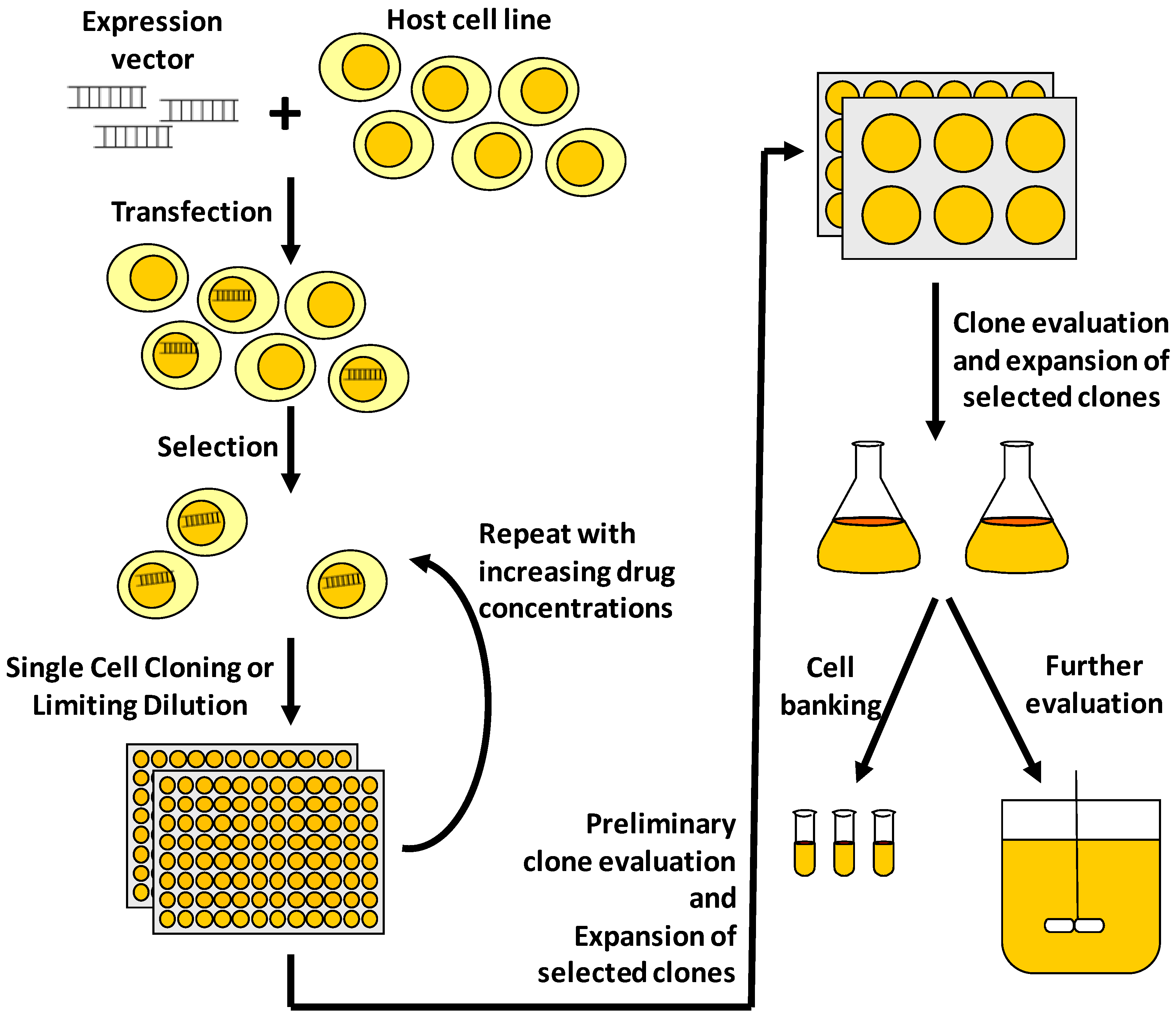
Pharmaceuticals Free Full Text Advances In Mammalian Cell Line Development Technologies For Recombinant Protein Production

Custom Cell Line Development

Cell Lines Glycotope

Stable Cell Line Development Service Constitutive Expression Trenzyme Com

Towards Next Generation Cho Cell Line Development And Engineering By Systems Approaches Sciencedirect
Cell Line Development Services Cho Hek Excellgene

Which Platform Partner For Cho Cell Line Development Gtp Technology



